Recent advances in neuroscience and neurotechnology promise restoration of functional sight to the blind through sending signals straight to the brain’s visual cortex. Similar to cochlear implant that has done wonders for the deaf, research is under way to restore visual perception to the blind through an implant that work by stimulating the visual cortex as it tries to recreate the activity hitherto triggered by the optic nerve.
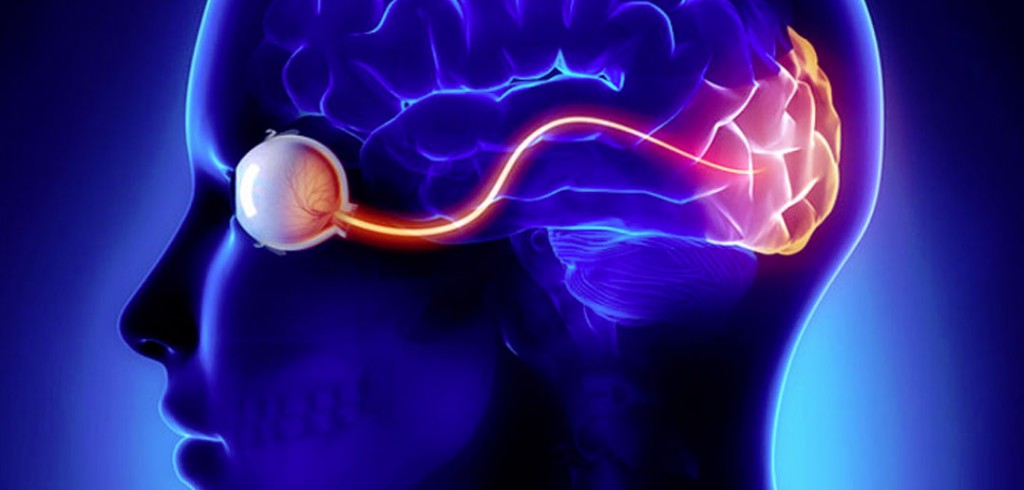 The implant is designed to create the sensation of seeing even without actual input from the eyes. Transmitting video images from a camera directly to the brain, the device targets the brain instead of the eyes, giving blind patients the ability to make partial sense of their surroundings.
The implant is designed to create the sensation of seeing even without actual input from the eyes. Transmitting video images from a camera directly to the brain, the device targets the brain instead of the eyes, giving blind patients the ability to make partial sense of their surroundings.
As a matter of fact, visual prosthetics have undergone rapid advancement since their early prototypes. They have the capability of artificially encoding sensation into the brain to produce spots of light, or phosphenes, in one’s visual field.
How vision-assisting implants work
To put it simply, visual prosthetics technology works in the following ways to restore vision, however fractional, to the blind:
- A camera embedded in a pair of thick, black-rimmed glasses records patient’s field of view.
- This camera, connected to a computer, sends a live video that the computer translates into electronic signals.
- These signals are then sent via a port surgically embedded in the backside of the skull. That port connects to a 100-electrode implant in the visual cortex of the brain.
- The implant stimulates neurons in patient’s visual cortex.
- Subsequently, the patient experiences a low-resolution perception of the surroundings in the form of white-yellow dots and shapes called phosphenes that can be interpreted as objects.
Evolution of vision-restoration technology
The focus on earlier research in bioengineering technologies had been on creating an artificial eye or retina. Not long ago, the Artificial Retina Project brought together a number of research institutions to develop artificial retinas to help the blind see. The work resulted in the Argus systems which helped for those suffering from retina-destroying diseases. These systems involved the use of a camera mounted on glasses; a computer to convert sensory data, and an implant with an array of electrodes embedded in the retina (instead of the brain). Over the years, Argus I and Argus II systems conducted human trials, and received approval in Europe in 2011 and the US in 2013 to sell their bionic eyes to eligible individuals.
The implant comprised single sheet of retinal pigment epithelial (RPE) cells derived from human embryonic stem cells. However, attempts to create a bionic eye focused on implanting into the eye itself. They depended on the need of an eye with a working optic nerve. But, for many people, whose blindness originates beyond the retina, the technology of artificial eye falls short of solving their blindness. These people suffer from damage to the nerve system connecting the retina to the back of the brain or the neural pathways between eyes and brain.
Headway in brain implants
Brain-Machine Interface (BMIs) is quickly advancing on a number of fronts such as helping paralyzed folks control robotic arms and typing messages with just their thoughts. Companies working in the BMIs domain have pivoted from artificial retinas to the brain itself. They have tested a system that skips the retina and sends visual information straight to the brain.
Under this system, as mentioned earlier, a feed from a video camera mounted on dark glasses is converted to electric pulses sent to an implant that stimulates the brain. Subsequently, the user sees a low-resolution pattern of phosphenes or white dots on a black background.
Utah array, a widely used brain implant, is a few millimetres wide square array that contains 100 electrode spikes which are inserted into the brain. Each spike stimulates neurons in the brain. The array is just smaller than the raised tip on the positive end of an AAA battery. Tiny electrode spikes, each about a millimetre tall—look like a miniature bed of nails. Each spike is capable of delivering a current to one-four neurons.
The implant that transmits video images directly to the brain is indeed a breakthrough. The video camera attached to glasses sends footage to electrodes implanted in the visual cortex of the brain, enabling participants to experience some level of vision where none had previously been possible.
The technology is remarkable in that it bypasses both the eye and the optic nerve which would normally transmit sensory information to the brain.
A case study in brain implant
Neuroengineer Eduardo Fernandez, director of neuroengineering at the University of Miguel Hernandez in Spain, recently made news by empowering a blind woman with rudimentary vision by sending visual information straight to her brain.
The patient in question, 57-year-old Bernadeta Gomez, had been totally blind since she was 42 when she started suffering from toxic optic neuropathy, a condition that totally severed the connection between her eyes and her brain.
Fernandez implanted a tiny chip into Gomez’s brain, containing 100 millimetre-long electrodes that stimulated specific neurons that allowed her to see a rough image of the world for the first time in 16 years. She saw the world through phosphenes – an experience of seeing light without being triggered by retinal stimulation, it gave her a faint perception of the shapes and space around her at an approximate resolution of 10×10 pixels.
To compensate for poor resolution, Fernandez incorporated facial recognition software that helped her associate the set of corresponding phosphenes that she received through the device with specific people. The device included a belt with a button to amplify dark objects in the sun or light objects in the dark.
The system allowed the woman to have “rudimentary” vision despite destruction of the bundles of nerves that connected her eyes to her brain. She was able to see a low-resolution version of what the world looks like. The glasses allowed her to identify letters, lights and people. It came out looking like glowing whitish-yellowish dots and shapes, but it was at least a semblance of the world rather than pure darkness.
Challenges in sight restoration
The implant tends to decay due to natural bodily processes. The body’s immune system starts to break down the electrodes that damage or scar the surrounding brain tissue. Scarring eventually weakens the signal and the electrodes can only interact with a handful of neurons, eventually rendering them useless. The prosthesis can be left in the brain for a short period of time as nobody knows how long the electrodes can last without degrading either the implant or the user’s brain.
Another downside is that when the implant is inserted, the electrodes pierce the surface of the brain; when it’s removed, 100 tiny droplets of blood form in the holes.
The road ahead
Fernandez’s research points the way to other ambitious projects aiming to bridge the gap between computers and humans using brain-computer interfaces (BCI). Significantly, BCI aims to change the quality of life for people with disabilities. Britain’s Royal Society has called for more attention to be paid to the development and regulation of BCI, saying the technology has the potential to usher in a “new and collaborative form of intelligence”.
Fernandez’s process also relies on more than just the hardware. It deploys machine learning to write the software that translates visual information into neural code that can be further refined. In future, as scientists work on brain implants as a whole, the components will no doubt improve in parallel.
Ferndandez believes his implant can be modified to last longer, and while the current maximum resolution is 10 by 10 pixels, he envisions to achieve a resolution of at least 60 by 60 pixels by implanting as many as 6 electrodes on each side of the brain.
Researchers from Harvard Medical School have been working with a new kind of implant which is unaffected by scar tissue. Doing away with the need of penetrating the organ, these new electrodes are placed beneath the skull to rest on the surface of the brain. Moreover, powerful magnetic fields are used instead of electrodes to induce brain activity.
The ambitious Neuralink project of Elon Musk, CEO of SpaceX and Tesla, aims at developing implants threading directly onto neurons and providing two-way communication between the brain and a smart-phone app. He wants to bring computing to people with severe loss of brain function. To achieve better precision, he wants the surgical implants to be non-major and completed by a robot. Soft, thread-like electrodes are being developed by Neuralink to be able to deftly lace them into brain tissue by a robot. The organization is aiming to include 3,000 electrodes on their device to chat up far more neurons than is currently possible.
In fact, more and more new technologies are in the works in the field of visual prostheses. Usually, the sight-giving glasses connect to power, computer and implant via cables. The ideal device needs to be wireless so that it can last for a long time—limiting the number of surgeries needed—and offering greater precision and resolution. It could do away with electrodes altogether, using light or chemicals to control gene-edited neurons.
Filed Under: ARM., Blog entry, Featured, Tech Articles


Questions related to this article?
👉Ask and discuss on Electro-Tech-Online.com and EDAboard.com forums.
Tell Us What You Think!!
You must be logged in to post a comment.