In the previous article, we discussed different types of power supplies. For prototyping and testing circuits, the linear regulated power supply is the most economical and feasible do-it-yourself solution. The power output from a linear regulated supply can also be adjusted with the help of a variable resistor. Electronics hobbyists and beginners who do not want to take pain in designing their own power supply can buy an adjustable bench power supply for regular circuit testing and prototyping. Most of the embedded systems are battery-based, so sooner or later, you will need to add a battery-based power supply to your designs (circuits). Even circuits that are not entirely battery-based may be using batteries for power backup. So, it is essential to learn about these electrochemical components before anything else, as these will be making your circuits live in most of the situations.
What are batteries
Batteries are electrochemical devices that store electrical energy in the form of chemical energy. When they are connected in a circuit, current starts flowing across their terminals as chemical energy stored in them is discharged back as electrical energy. Supercapacitors are different from batteries as they store electrical energy as an electrostatic field while batteries store energy electrochemically.
Batteries are two-terminal devices and always have a positive (anode) and a negative terminal (cathode). They are made up of three components – anode, cathode, and electrolyte. When a battery is connected to a circuit, electrons flow from cathode to anode due to chemical reactions with the electrolyte. Obviously, the direction of the conventional current is from anode to cathode.
Types of batteries
There are four main types of batteries:
- Primary Batteries
- Secondary Batteries
- Reserve Cells
- Fuel Cells
Primary batteries
Primary cells and batteries are non-rechargeable and disposable batteries. The electrochemical reactions in these batteries cannot be reversed, so they cannot be recharged once depleted and can even discharge if not used for a long time. These batteries are meant for single use and need to be discarded once depleted. Such batteries are most suitable for battery-based applications, which usually have low power requirements and are meant to run entirely by battery supply for a long time. Primary batteries are also used in many military-grade applications (radio and communication) as it is convenient to replace batteries than recharge them in such applications. Commonly used primary batteries include coin cells, AA, and AAA batteries.

The RJD Series lithium-ion coin-cell batteries feature the high power density required in IoT wearable applications. (Image: Illinois Capacitor)
These low cost, small size batteries are usually dry, have good charge retention, and are meant for long-term use. They are not designed for nor are suitable for heavy load applications where a high discharge rate is required, nor are they suitable for power backup or load leveling.
There are many types of primary batteries available. Some of the popular primary batteries include the following.
Alkaline Batteries: These are the most popular primary batteries and comprised of 1.5V cells. These have high specific energy, a long life, are eco-friendly, safe to use, leak-proof, and low cost. These are ideal for low current applications and are used in many consumer devices like TV remotes, wall clocks, and toys. Their main downside is weight as they are heavy.
Zinc Carbon Batteries: These are heavy-duty batteries and are a low-cost alternative to alkaline batteries. However, they have lower specific energy (energy density) and poor discharge rate performance. These are generally used as a cost-cutting replacement of alkaline batteries.
Primary Lithium Batteries: These small-size and lightweight batteries are intended to be used for low current applications. These are used as an alternative to alkaline batteries, where size and weight are a prime concern against the cost.
Mercury Batteries: These are not dry batteries like other primary battery chemistries. These small size batteries have a longer life and a stable voltage. They are generally used in size constraint applications.
Secondary batteries
Secondary batteries are rechargeable batteries. Their electrochemical reactions can be reversed by recharging them. In the process of recharging, charging current is passed across their terminals opposite to the direction of discharging current. Once recharged, these batteries are restored to their pre-discharge state.
Secondary batteries have a high initial cost; however, as they can be reused many times, they have lower life-cycle costs. In many applications, it is more cost-effective to use low-power secondary batteries than primary batteries for long time usage. That is why low power secondary batteries are commonly used in mobile devices and gadgets. These batteries are also useful in heavy-duty applications and those applications where high discharge rate performance is required. It is the reason that secondary batteries are used for applications like load leveling, electric vehicles, and power backup. Some of the popular secondary batteries include the following:
Lithium-Ion Batteries: These are the most popular secondary batteries. They come in a variety of chemistry like lithium cobalt oxide, lithium-sulfur, and lithium iron phosphate. These lightweight batteries have the highest energy density and excellent discharge rate performance and are widely used in smartphones, portable gadgets, and battery-based consumer devices. Recently, a Nobel prize in chemistry for 2019 was awarded to three scientists — John B Goodenough, M Stanley Whittingham, and Akira Yoshino — for their work in developing lithium-ion batteries.

A pin‐type Lithium‐ion battery suitable for powering small applications such as wearable devices. (Image: Panasonic Industrial Battery)
Lead Acid Batteries: These are the earliest type of secondary batteries. These are heavy, large-sized batteries that are used for heavy-duty applications with a lower cost than other batteries making them useful in non-portable applications such as n automobiles and for power backup. Despite low specific energy and heavy weight, these batteries have a high power to weight ratio. They come available as Sealed Lead Acid (SLA) batteries.
Nickel-Cadmium Batteries: These durable secondary batteries use metallic cadmium and nickel oxide hydroxide as electrodes and are known to deliver their full rated voltage at high discharge rates. These have a low self-discharge and long life. A major disadvantage of Ni-Cd batteries is that they have a memory — if they are not appropriately recharged, their life can shorten.
Nickel Metal Hydride: These batteries are similar to Ni-Cd batteries except that these use a hydrogen-absorbing alloy instead of cadmium. They have higher energy density and do not require a specific recharging procedure. However, these batteries have a short life compared to Ni-Cd and may need to be disposed of before a thousand cycles.
Reserve cells
Like primary batteries, reserve cells are also mostly for one-time usage but feature an electrolyte isolated from the electrodes or electrodes isolated from the electrolyte. Most of the reserve cells have isolated electrolytes. The electrolyte is activated only when the battery is used. As electrolyte and electrodes remain isolated, these batteries do not self-discharge (like primary batteries) and have a longer life. Some of the popular reserve cells include electrolyte activated batteries, heat-activated batteries (thermal batteries), water activated batteries, and gas activated batteries. These batteries are generally used in military-grade applications.
Fuel cells
In fuel cells, the active material is fed into the battery from outside. The battery remains operational only until active material is fed to it. Fuel cells are generally used in space applications but also in applications such as electric vehicles, load leveling, and power backup.
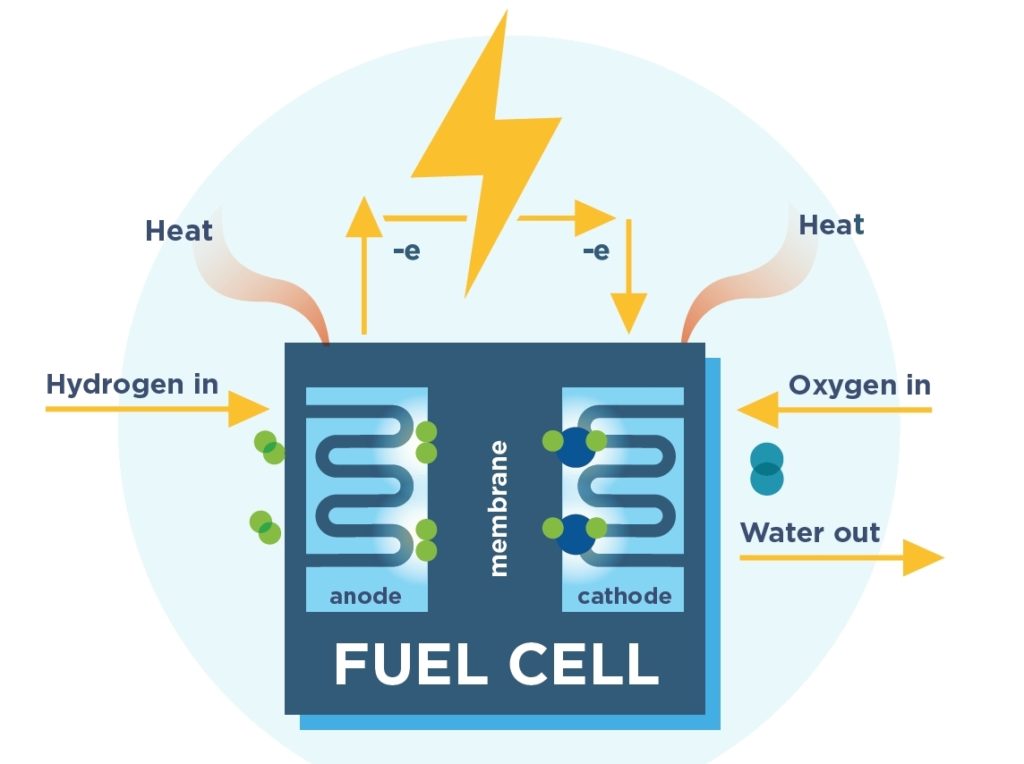
How a basic fuel cell works. (Image: FCHEA)
Most battery-based embedded devices use primary batteries. Many embedded devices may use secondary batteries such as in industrial applications, where using secondary batteries may be much cost-effective. Reserve cells and fuel cells have limited applications and may not be seen in widely used electronics. Like inductors, batteries are specific to applications. So, it is not a daunting task to choose a battery type for a given circuit or application. But you do need to consider other requirements such as the terminal voltage, safety, compliance, size, and geometry in selecting a battery for a given application.
We have already discussed different types of batteries based on reusability and their chemistry. In the next article, we will talk about the geometry and size of batteries.
You may also like:
Filed Under: Tutorials







Questions related to this article?
👉Ask and discuss on Electro-Tech-Online.com and EDAboard.com forums.
Tell Us What You Think!!
You must be logged in to post a comment.